|
This linker does not have any repeats that can lead to homologous recombination. The most widely-used flexible linker has the sequence of (Gly-Gly-Gly-Gly-Ser)n. Primarily, stretches of Gly and Ser residues (“GS” linker) are used to improve folding and stability of a fusion protein. These residues provide a certain degree of movement. Researchers often use flexible linkers containing small non-polar glycine (Gly) or polar serine (Ser) residues. Direct fusion of the protein and tag (especially large protein tag) domains without a linker may lead to undesirable outcomes, such as misfolding of the fusion proteins, low protein yield, or impaired function. Selection of a suitable linker sequence to join the target protein and the epitope tag is an important aspect of a recombinant fusion. Depending upon the requirements of the experiment, epitope tags can also be introduced internally in the target gene, if necessary. There is minimal risk of interference with protein function and conformation if the tag is introduced at the N- or C- termini, unless either extremity is known to have a specific function. Positioning of the tag: Epitope tags are commonly positioned near the end of the N-terminus or C-terminus of a target protein.Such aberrant transcripts are degraded and have no expression of desired protein. 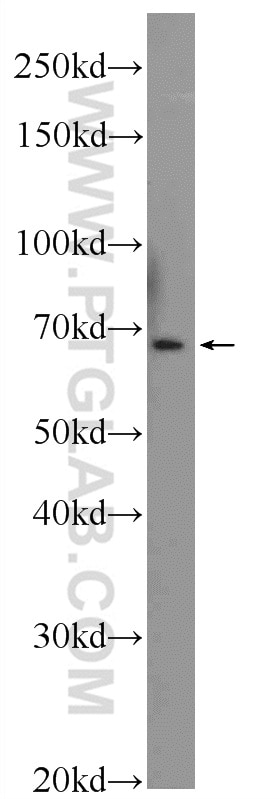
This can lead to stalling of the ribosome at the end of the transcript. Without a stop codon, the signal to remove the ribosome from the transcript is missing.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
